Baby Losing Weight Low Level High Potassium Level
- Example report
- Open up Access
- Published:
Early-onset neonatal hyperkalemia associated with maternal hypermagnesemia: a case report
BMC Pediatrics volume 18, Article number:55 (2018) Cite this article
Abstract
Background
Neonatal nonoliguric hyperkalemia (NOHK) is a metabolic abnormality that occurs in extremely premature neonates at approximately 24 h later birth and is mainly due to the young functioning of the sodium (Na+)/potassium (K+) pump. Magnesium sulfate is frequently used in obstetrical practice to forestall preterm labor and to treat preeclampsia; this medication tin also cause hypermagnesemia and hyperkalemia by a mechanism that is dissimilar from that of NOHK. Herein, we report the first instance of very early-onset neonatal hyperkalemia induced by maternal hypermagnesemia.
Case presentation
A neonate born at 32 weeks of gestation developed hyperkalemia (G+ half dozen.4 mmol/50) 2 h afterwards birth. The neonate'southward blood potassium concentration reached vii.0 mmol/L 4 h after nativity, despite good urine output. The neonate and his female parent had severe hypermagnesemia caused by intravenous infusion of magnesium sulfate given for tocolysis due to pre-term labor.
Decision
The early on-onset hyperkalemia may have been caused past the accumulation of potassium ions transported through the placenta, the shift of potassium ions from the intracellular to the extracellular space in the babe due to the malfunctioning of the Na+/K+ pump and the inhibition of renal distal tube potassium ion secretion, there is a possibility that these mechanisms were induced by maternal and fetal hypermagnesemia afterward maternal magnesium sulfate administration. Because neonatal hyperkalemia poses a significant adventure for the development of life-threatening cardiac arrhythmia, this instance highlights the necessity of maternal blood magnesium monitoring during magnesium sulfate assistants and neonatal blood potassium monitoring when at that place is astringent maternal hypermagnesemia at delivery.
Background
Neonatal nonoliguric hyperkalemia (NOHK) is a frequently observed electrolyte imbalance that occurs in nonoliguric premature infants during the first days after birth [ane]. NOHK results secondary to potassium shift from the intracellular infinite to the extracellular space partly because of the malfunctioning of the Na+/One thousand+ pump due to the immature activeness of Na+/Thou+-ATPase [2]. It tin can cause life-threatening cardiac arrhythmia as a severe complication [3].
Magnesium is a modulator of the Na+ and M+ ion transport systems in numerous tissues, and hypermagnesemia inhibits Thou+ ion transport from the extracellular to the intracellular infinite through the Na+/One thousand+ pump [iv]. Additionally, hypermagnesemia inhibits renal distal tube Thou+ ion secretion by the renal outer medullary Yard+ (ROMK) aqueduct, which is an inward-rectifying Thousand+ ion channel responsible for basal K+ ion secretion [v, six]. An overdose of magnesium sulfate, frequently used in obstetrical practice for the prevention of preterm labor and to treat preeclampsia, can crusade maternal hypermagnesemia [7]. Because the magnesium ion administered to the mother readily crosses the placenta, infants born to mothers with hypermagnesemia often develop transient hypermagnesemia during the first days afterwards nativity [viii]. Moreover, it has been reported maternal and neonatal magnesium concentrations were highly correlated [9].
Although transient hyperkalemia during magnesium sulfate therapy in two pregnant drug abusers has been reported [7], there has been no report on neonatal transient hyperkalemia acquired by maternal magnesium sulfate therapy. Here, we nowadays the case of a male person infant at 32 weeks gestation (weight 1268 grand) that developed hyperkalemia immediately afterwards birth due to neonatal and maternal transient hypermagnesemia afterwards administration of magnesium sulfate.
Case presentation
A 29-year-old Japanese woman in her 2d pregnancy was being treated for pregnancy-induced hypertension and preeclampsia with a baste infusion of magnesium sulfate at 30 weeks 2 days gestation. The dose of continuously infused magnesium sulfate was 0.1 grand/h on day i, 0.5 g/h on day ii–v, and ane.0 g/h on day 6–12. The dose was increased to two.0 g/h to control an imminent preterm delivery on day 12, just before an emergency transfer to our infirmary. During this treatment, maternal serum magnesium levels were not monitored. The mother had no medical conditions or any intake of other medications inside these 12 days, which could have influenced blood potassium concentration.
On admission to our hospital, the patient could non mobilize independently because of hypotonia caused past astringent hypermagnesemia and hypocalcemia (Table 1). On the same day, she delivered a male infant through vaginal birth. Her laboratory analysis at vii h before and seven h afterwards delivery showed that the serum concentration of potassium, sodium, and creatinine ranged from 4.3 to half-dozen.0 mmol/L, 133 to 128 mmol/L, and 0.85 to 0.95 mg/dL, respectively. These levels returned to inside normal ranges at v days after delivery.
The male infant was born at 32 weeks gestation weighing 1268 m and with Apgar scores of viii at 1 min and ix at v min. He was immediately admitted to the neonatal intensive care unit in our hospital. There was no evidence of respiratory distress syndrome on chest radiograph, and he had a stable microbubble test. His heart rate was 130 beats per minute, and the arterial blood pressure was 42/22 mmHg, with normal contraction of the left ventricle confirmed by echocardiographic examination. His muscle tone was adamant to be inside the normal range by two expert neonatologists. The size and shape of both kidneys on ultrasonography were within the normal range, and his first urine was observed at 2 h afterwards birth.
Laboratory data showed that his venous blood potassium concentration was 6.iv mmol/50 at two h after nativity and reached 7.0 mmol/L at 4 h after nativity fifty-fifty though he had sufficient urine output (Table one, Fig. i). Glucose-insulin combined therapy was administered for 54 h with an insulin infusion rate betwixt 0.9 units/kg/day and 1.9 units/kg/twenty-four hours to maintain his serum potassium concentration below 6.0 mmol/Fifty. All blood samples were obtained from a catheter inserted directly into the correct radial artery. During the therapy, he was not given potassium. His urine output was 4.one mL/kg/h during the kickoff eight h, half dozen.two mL/kg/h during the next twenty-four hour period, and 3.3 mL/kg/h during the third twenty-four hour period after birth with insufficient urinary potassium excretion (Table i).
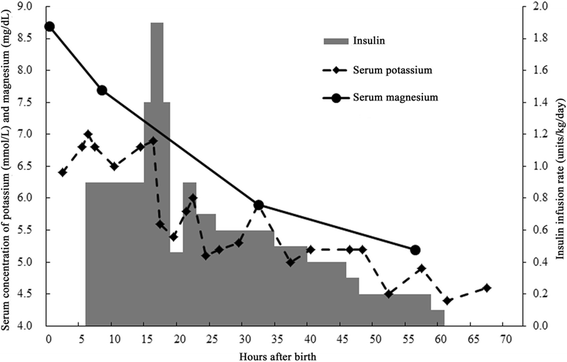
The neonate'due south clinical course until 72 h later on birth. The clinical course of the patient is shown. The main indicators include the serum potassium and magnesium concentrations and infusion charge per unit of insulin until 72 h afterwards birth
Laboratory analysis of his umbilical blood besides showed hypermagnesemia at birth, which gradually subsided (Table 1, Fig. 1). The serum calcium concentration remained over vii.3 mg/dL with intravenous baste assistants of calcium sulfate. Although transient hyponatremia was observed at half-dozen h after nascency, information technology recovered in 24 h without sodium administration.
After resolution of NOHK, the neonate's serum concentrations of potassium and magnesium were within the normal range. He was discharged from our infirmary at 72 days subsequently nascence with no clinical complications and had normal growth and development at 3 years of historic period.
Word
NOHK is a metabolic disorder that usually occurs in extremely premature neonates; therefore, the onset at 32 weeks old in the neonate presented in this example is less expected. Furthermore, the clinical course in this case was very unique in terms of the early onset at 2 h after nascency and prolonged for 54 h nether glucose-insulin combined therapy.
One possible explanation as to the cause of the early onset hyperkalemia is maternal hyperkalemia caused by hypermagnesemia [7]. Usually, the fetal plasma M+ ion concentration is higher than the maternal plasma concentrations [10] because of active One thousand+ ion ship across the placenta [eleven]. In this instance, the maternal potassium concentration increased from 4.3 mmol/L at vii h earlier commitment to 6.0 mmol/L at 7 h after delivery. Information technology is therefore possible that the backlog K+ ions might have crossed from the mother to the fetus through the placenta and might accept partially contributed to early onset neonatal hyperkalemia.
Alternatively, the prolonged hyperkalemia may have been secondary to a shift in Thousand+ ions from the intracellular to the extracellular space. In premature infants with NOHK, erythrocyte Na+/K+-ATPase activity is significantly lower and the malfunctioning of the Na+/Yard+ pump induces a G+ ion shift from the intracellular to the extracellular infinite [1, 2]. This case may illustrate that fifty-fifty in an infant at 32 weeks gestation, Na+/Thousand+-ATPase action may be immature and outcome in malfunctioning of the Na+/K+ pump. In addition to prematurity, neonatal hypermagnesemia may produce a Chiliad+ ion shift from the intracellular to the extracellular space because of the loftier concentration of magnesium ions, which modulates the Na+/1000+ ion transport systems in numerous tissues and inhibits the Na+/Yard+ pump commutation [4].
An boosted machinery whereby neonatal hypermagnesemia results in hyperkalemia is through the inhibition of renal distal tubule K+ ion secretion. Magnesium is idea to inhibit renal distal tube K+ ion secretion by suppressing the ROMK channel, which is an inward-rectifying K+ ion channel responsible for basal K+ ion secretion [5, 6]. In the neonate described in this case, post-delivery urine output was sufficient; however, the fractional excretion of potassium (FeK) at 0, i, and 2 days after birth was 36.5, 24.9, and four.4%, respectively, and these levels of excretion are lower than the average FeK (twoscore ± 5%) in NOHK during the outset few days after birth [12]. The amount of urinary excretion of potassium is larger in an infant with NOHK than an babe with normokalemia [thirteen]. However, in this example, information technology is interesting that urinary excretion of potassium was suppressed despite hyperkalemia, because of hypermagnesemia.
Thus, this progressive early-onset hyperkalemia may be caused by maternal and fetal hypermagnesemia. The underlying mechanism of this hyperkalemia is mainly causeless to be secondary to hypermagnesemia and subsequent malfunctioning of the Na+/K+-ATPase and inhibition of secretion in the ROMK channel.
Decision
Maternal and fetal hypermagnesemia tin can induce quickly progressive hyperkalemia in neonates. Because hyperkalemic infants are at high risk of developing life-threatening cardiac arrhythmias, we highlight the necessity of maternal blood magnesium monitoring during intravenous infusion of magnesium sulfate for tocolysis, likewise as neonatal blood potassium monitoring when there is maternal astringent hypermagnesemia at commitment.
Abbreviations
- NOHK:
-
Neonatal nonoliguric hyperkalemia
- ROMK:
-
Renal outer medullary Grand+
References
-
Sato K, Kondo T, Iwao H, Honda Due south, Ueda K. Internal potassium shift in premature infants: crusade of nonoliguric hyperkalemia. J Pediatr. 1995;126:109–13.
-
Baumgart S. Astute problems of prematurity: balancing fluid volume and electrolyte replacements in very low birth weight (VLBW) and extremely depression birth weight (ELBW) neonates. In: Oh W, Guignard JP, Baumgart South, editors. Nephrology and fluid/electrolyte physiology. Philadelphia: Saunders; 2008. p. 161–83.
-
Hu PS, Su BH, Peng CT, Tsai CH. Glucose and insulin infusion versus kayexalate for the early treatment of not-oliguric hyperkalemia in very-depression-nativity-weight infants. Acta Paediatr Taiwan. 1999;40:314–viii.
-
Bara K, Guiet-Bara A, Durlach J. Regulation of sodium and potassium pathways by magnesium in prison cell membranes. Magnes Res. 1993;six:167–77.
-
Heller BI, Hammarsten JF, Stutzman FL. Concerning the furnishings of magnesium sulfate on renal function, electrolyte excretion, and clearance of magnesium. J Clin Invest. 1953;32:858–61.
-
Huang CL, Kuo E. Mechanism of hypokalemia in magnesium deficiency. J Am Soc Nephrol. 2007;18:2649–52.
-
Spital A, Greenwell R. Astringent hyperkalemia during magnesium sulfate therapy in 2 meaning drug abusers. South Med J. 1991;84:919–21.
-
Rantonen T, Kääpä P, Jalonen J, Ekblad U, Peltola O, Välimäki I, et al. Antenatal magnesium sulphate exposure is associated with prolonged parathyroid hormone suppression in preterm neonates. Acta Paediatr. 2001;xc:278–81.
-
Sherwin CM, Balch A, Campbell SC, Fredrickson J, Clark EA, Varner K, et al. Maternal magnesium sulphate exposure predicts neonatal magnesium blood concentrations. Basic Clin Pharmacol Toxicol. 2014;114:318–22.
-
Shennan DB, Boyd CA. Ion transport by the placenta: a review of membrane send systems. Biochim Biophys Acta. 1987;906:437–57.
-
Ashoor IF, de Jesús-González N, Somers MJ. Fluid and electrolyte physiology in the fetus and neonate. In: Chishti AS, Alam South, Kiessling SG, editors. Kidney and urinary tract diseases in the newborn. Berlin Heidelberg: Springer; 2014. p. 77–98.
-
Omar SA, DeCristofaro JD, Agarwal BI, LaGamma EF. Effect of prenatal steroids on potassium balance in extremely low nascency weight neonates. Pediatrics. 2000;106:561–seven.
-
Stefano JL, Norman ME, Morales MC, Goplerud JM, Mishra OP, Delivoria-Papadopoulos M. Decreased erythrocyte Na+, K+ -ATPase activity associated with cellular potassium loss in extremely low birth weight infants with nonoliguric hyperkalemia. J Pediatr. 1993;122:276–84.
Acknowledgements
The authors would like to thank Dr. Endo for thoughtful comments during the writing of this manuscript.
Funding
This work was non funded past institutional, financial or textile support from external sources.
Availability of information and materials
The datasets used and/or analyzed during the current study are available from the respective author on reasonable asking.
Author data
Affiliations
Contributions
KT reviewed the literature and prepared the manuscript. HMo and SM helped to typhoon the manuscript. RS was involved in collection of the clinical details. HMi and NK assisted with manuscript revision. MI reviewed the literature, prepared the manuscript, and is the corresponding author. All authors read and approved the last version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All examinations and investigations in this instance were approved by the IRB of Kumamoto University (registration No. 1207).
Consent for publication
The mother provided written informed consent for her and her son's details to exist published in this case written report.
Competing interests
The authors declare that they have no competing interests.
Publisher'due south Notation
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Admission This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original writer(southward) and the source, provide a link to the Creative Eatables license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/null/one.0/) applies to the data fabricated available in this commodity, unless otherwise stated.
Reprints and Permissions
About this article
Cite this article
Tanaka, K., Mori, H., Sakamoto, R. et al. Early-onset neonatal hyperkalemia associated with maternal hypermagnesemia: a case report. BMC Pediatr 18, 55 (2018). https://doi.org/10.1186/s12887-018-1048-4
-
Received:
-
Accepted:
-
Published:
-
DOI : https://doi.org/10.1186/s12887-018-1048-four
Keywords
- Neonatal nonoliguric hyperkalemia
- Progressive early-onset hyperkalemia
- Maternal hypermagnesemia
- Na+/1000+-ATPase
- Renal outer medullary potassium channel
Source: https://bmcpediatr.biomedcentral.com/articles/10.1186/s12887-018-1048-4
0 Response to "Baby Losing Weight Low Level High Potassium Level"
Post a Comment